The pH Balancing Act in Mycorrhiza: How to Maintain Optimal pH for Maximum Effectiveness
Why pH Matters More Than You Think?
In today’s shift toward sustainable agriculture, mycorrhiza biofertilizers are playing a vital role in improving soil health and crop productivity. However, one critical factor that often goes unnoticed is pH stability.
For Glomus species—the most widely used arbuscular mycorrhizal fungi—pH directly impacts:
- Spore viability
- Root colonization
- Nutrient uptake efficiency
- Shelf life of the product
This is why India’s FCO standards for biofertilizers clearly define a specific pH range for commercial products. Simply put, if the pH is not right, the performance of even the best formulation can decline.
Understanding the Role of pH in Mycorrhiza
The success of mycorrhizal biofertilizers depends largely on the development of the extraradical mycelium—the fungal network responsible for nutrient absorption.
When the pH is within the ideal range for mycorrhiza growth, the fungus actively expands this network, improving the plant’s access to phosphorus and micronutrients. However, under low pH conditions, this growth is restricted, reducing the overall effectiveness of the biofertilizer.
In simple terms, pH determines whether the fungus just survives—or truly performs.
Why pH is Not Constant?
One of the biggest challenges in maintaining biofertilizer pH stability is that pH keeps changing at different stages—from production to field application.
1. Fungal Nutrient Uptake
The fungus actively modifies its local pH depending on the form of nitrogen available. When supplied with nitrate, the extraradical hyphae of Glomus intraradices strongly increased the pH of the medium. This occurs because the fungus takes up nitrate through a mechanism coupled with proton (H⁺) co-transport, effectively removing acid from its surroundings and causing the pH to rise.
In contrast, when nitrogen sources differ, the pH may decrease.
This nutrient-driven pH adjustment is a physiological adaptation: the fungus changes its local chemistry to optimize nutrient capture. This natural response helps optimize nutrient absorption, but it also leads to pH fluctuations within the system.
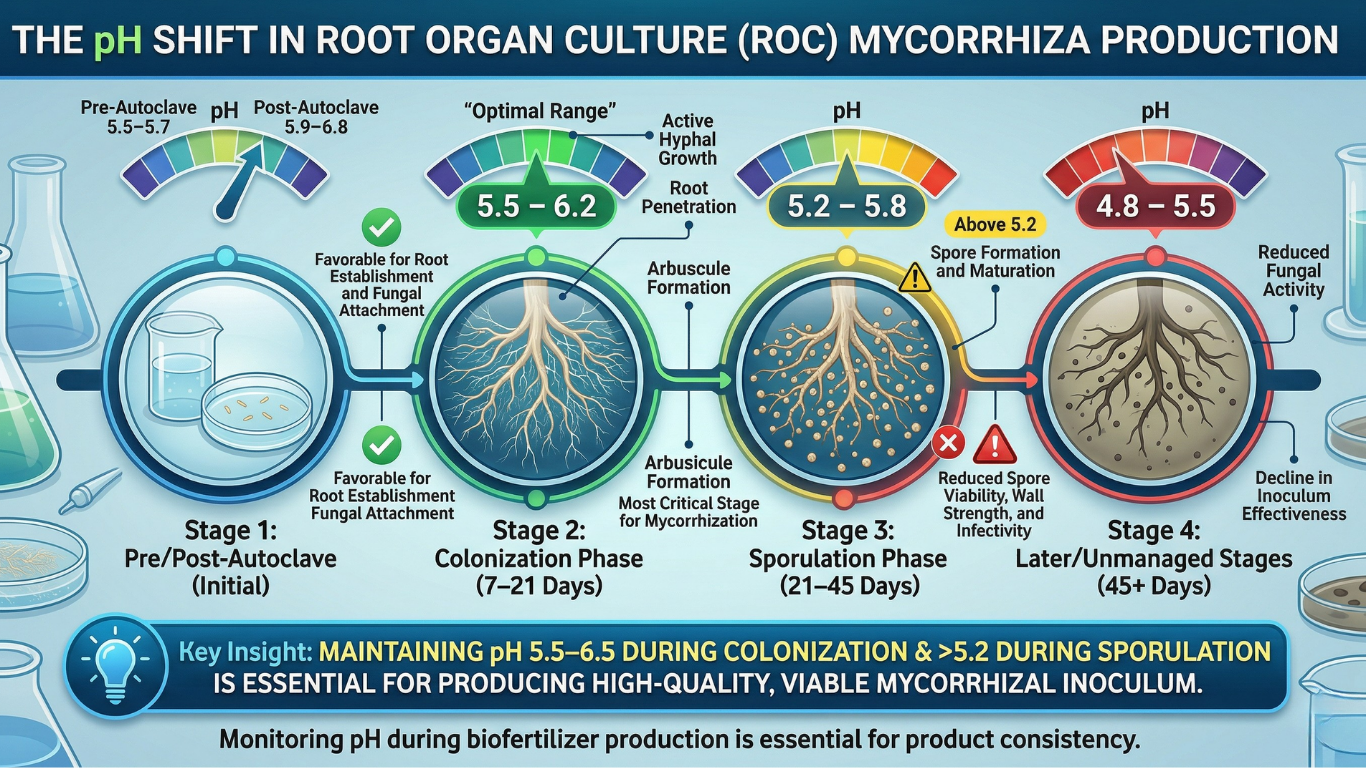
2. pH Shift in ROC During Manufacturing: Why It Matters
In Root Organ Culture (ROC)-based mycorrhiza production, pH does not remain constant—it shifts at every stage due to media chemistry, root metabolism, and fungal activity. These changes directly influence mycorrhization efficiency, fungal viability, and sporulation capacity.
The process typically begins with a pre-autoclave pH of 5.5–5.7, which increases to around 5.9–6.8 after autoclaving due to heat-induced chemical reactions. This slightly higher pH is generally favorable for initial root establishment and fungal attachment.
During the active colonization phase (7–21 days), pH gradually declines to 5.5–6.2 as roots and fungi release organic acids. This range is considered optimal for hyphal growth, root penetration, and arbuscule formation, making it the most critical stage for effective mycorrhization.
As the culture progresses into the sporulation phase (21–45 days), pH further drops to 5.2–5.8, which supports spore formation and maturation. However, if pH falls below 5.0, it can negatively impact spore viability, wall strength, and infectivity, ultimately reducing product quality.
In later stages, unmanaged cultures may reach pH 4.8–5.5, leading to reduced fungal activity and decline in inoculum effectiveness.
Key Insight:
Maintaining pH within the range of 5.5–6.5 during colonization and above 5.2 during sporulation is essential for producing high-quality, viable mycorrhizal inoculum in ROC systems
This is why monitoring pH changes during biofertilizer production is essential for maintaining product consistency.
3. Soil and Root Interaction
Once applied to the field, the fungus interacts with plant roots, which release organic acids and other compounds. Research on coffee seedlings showed that mycorrhizae induced a higher pH, a lower exchangeable acidity, and higher values of exchangeable cations in the rhizosphere. This creates a feedback loop where the fungus modifies pH, which influences root activity, which in turn affects the fungus.
This interaction is key to the success of mycorrhiza in sustainable agriculture.
Choosing the Right Carrier for pH Stability
The carrier material—the physical matrix that houses the fungal propagules—significantly influences pH stability. Different carriers have different buffering capacities, meaning they vary in their ability to resist pH changes. An ideal carrier should not only buffer pH but also support microbial life and ensure effective delivery to plant roots.
- Clay-based carriers (Bentonite):Offer high buffering capacity and excellent pH stability. Their strong adsorption properties help resist pH fluctuations, making them one of the best carriers for mycorrhiza biofertilizer with longer shelf life.
- Peat-based carriers:Widely used but more prone to pH drift due to ongoing microbial activity and organic matter decomposition, which can gradually acidify the system.
- Vermiculite-based carriers:Provide good aeration and structure but have moderate buffering capacity. They are sensitive to processing conditions, especially autoclaving, which can cause noticeable pH shifts.
- Humic substance-based carriers (Humic acid / Potassium humate):Primarily act as protectants rather than structural carriers. They improve microbial survival under stress (temperature, drying, chemical exposure) and enhance formulation stability. However, their behavior is pH-dependent, as changes in pH alter their molecular structure, affecting their protective efficiency and colloidal stability.
- Dextrose-based carriers:Serve as a readily available energy source for the fungus rather than a buffering system. They support early microbial activation after application. These formulations are typically maintained within a pH range of 6.0–7.5, aligning well with optimal conditions for mycorrhizal activity, but require careful control as they offer limited pH buffering.
Key Insight:
While clay-based carriers provide the best pH stability, functional additives like humic substances and dextrose enhance microbial survival and activity. The most effective formulations often combine structural stability with biological support components.
Formulation Type and Its Impact on pH
The physical formulation of the product also affects pH stability. Four basic dispersal types are commonly produced: powder, granule, slurry, and liquid.
Powder Formulation
Powders provide quick root contact due to their fine particle size, but they are more sensitive to environmental changes and moisture, making them prone to pH drift.
Granular Formulation
Granules offer better protection to fungal propagules. Their larger size slows down chemical diffusion, helping maintain stable pH and improving shelf life.
Liquid Formulation
Traditional liquid formulations often have a shorter shelf life due to water-based instability. However, modern non-aqueous systems have significantly improved stability, reducing contamination risks and maintaining consistent pH over longer periods.
This is why formulation choice is critical for biofertilizer stability and performance.
Field Performance: What pH Range Actually Works?
After all the production and formulation considerations, the ultimate test is field performance. The available evidence indicates that Glomus species are adaptable across a wide soil pH range.
A glasshouse experiment examining Glomus fasciculatum across soil pH levels from 5.3 to 7.5 found that the fungus infected extensively at every level tested. However, the same study noted that total AM root colonization decreased at higher pH, and high pH also reduced arbuscule and vesicle formation. Different Glomus species also exhibit different pH optima for spore germination.
For most agricultural applications, a product pH in the 5.5–7.5 range will perform adequately in the field. However, for maximum functional benefit—particularly phosphorus acquisition—the evidence suggests a preference for the higher end of this range, where extraradical mycelium development is most robust.
Key Insights:
Functional range: 5.3 to 7.5 and Optimal range: 6.5 to 7.0
At higher pH levels, mycelial growth improves, enhancing nutrient uptake. However, extremely high pH can reduce the formation of important fungal structures.
Maintaining the ideal soil pH for mycorrhiza ensures consistent agricultural results.
The FCO Standard: pH 5.0–7.0 is it justified?
Yes. The Fertilizer Control Order (FCO) requirement of pH 6.5–7.0 for carrier-based products (and pH 5–7 for liquids) is supported by multiple lines of evidence.
- Spore germination: Research shows Glomus germinates best at pH 7, with maximum germination at this level.
- Fungal function: The Van Aarle study found that extraradical mycelium development – essential for nutrient and water uptake – is greater at higher pH.
- Species range: Different Glomus species have varying pH optima; the 6.5–7.0 range is a practical compromise that supports most commercially relevant species.
- Manufacturing reality: Autoclaving can shift pH significantly. Targeting 6.5–7.0 provides a buffer against drift during processing and storage.
- Liquid flexibility: The wider pH 5–7 range for liquid formulations acknowledges that non‑aqueous carriers have different chemistry and optimal ranges than solid carriers.
In short, the FCO standard aligns with both fungal biology and practical production constraints.
Key Takeaways for Better Results
To ensure maximum effectiveness of mycorrhiza biofertilizers, keep these points in mind:
- Maintain pH close to neutral (6.5–7.0)
- Choose clay-based carriers for better stability
- Prefer granular or advanced liquid formulations
- Monitor pH during production and storage
- Ensure compliance with FCO standards
Conclusion: pH is the Foundation of Performance
pH is not just a number on a specification sheet—it is the foundation of how effectively mycorrhiza biofertilizers work.
From production to field application, maintaining the correct pH ensures:
- Strong fungal growth
- Effective root colonization
- Improved nutrient uptake
- Better crop performance
For anyone involved in agriculture—whether manufacturer, agronomist, or farmer—understanding and maintaining the optimal pH for mycorrhiza is essential for achieving the best results.
The Clustered Code: Deciphering the Secret Life of Mycorrhizal Spores
If you’ve ever peeked through a microscope at arbuscular mycorrhizal (AM) fungi spores, you’ve likely seen them: not just solitary spheres, but intriguing clusters where spores appear glued together like microscopic bunches of grapes. For years, these aggregated spores—particularly of the ubiquitous species Glomus intraradices (now classified within the Rhizophagus genus)—were often counted as one unit or overlooked. But emerging science reveals that this "clustering" is not a random accident. It's a sophisticated survival strategy with profound ecological implications. Let's dive into the sticky, clever world of clustered endomycorrhizal spores.
The Microscopic Glue: How Spores Stick Together?
First, the mechanics. How are these spores so firmly aggregated?
The binding agent is a complex matrix of extraradical mycelial networks and glycoprotein-based secretions. Think of it not as a simple adhesive, but as a living, “biological hydrogel”.
- The Hyphal "Backbone": Often, spores are formed at the tips or along the length of the extraradical hyphae (the fungal network outside the plant root). Instead of detaching, the hyphae connecting them thicken, melanize, and persist, forming a durable structural scaffold that physically links the spores.
- The Secreted "Bio-Glue": As spores mature, they and their supporting hyphae exude a cocktail of glycoproteins, mucilaginous polysaccharides, and hydrophobins. This secretion hardens upon dehydration, forming a tough, protective crust that cements the entire assembly—spores, hyphae, and soil particles—into a single, cohesive unit. This crust is key to the cluster's resilience.
Why Cluster? The Ecological & Physiological Genius of Glomus intraradices
For Glomus intraradices, a champion of crop and grassland systems, spore clustering is a masterclass in evolutionary adaptation.
1. The Ecological - Propagule Bank Microsite:
- Desiccation Defence: The shared, hardened glycoprotein matrix significantly reduces the surface area exposed to the environment, minimizing water loss. It creates a buffered microclimate around the spores, protecting them from drought.
- Predator & Pathogen Deterrence: The same tough, often melanised crust that prevents water loss also acts as a physical barrier against grazing by soil micro arthropods and invasion by parasitic fungi. It's the spore's version of a castle wall.
- Nutrient Reservoir & Hub: A cluster isn't just spores; it's a package of stored carbon, lipids, and nutrients. This shared "communal pantry" may allow for resource redistribution, potentially aiding weaker spores within the unit. The interconnected hyphae can also function as a pre-established network, giving new germination an instant head start.
2. The Physiological Powerhouse:
- Synergistic Germination Signals: There is compelling evidence that spores in a cluster can engage in cross-talk. A spore initiating germination may release chemical signals (e.g., strigolactones in response to root exudates) that can stimulate or prime neighbouring spores in the cluster, leading to coordinated, potentially more effective colonization events.
- Adaptive Diversification (of Dormancy): Not all spores in a cluster germinate simultaneously. This asynchronous germination is a critical risk-management strategy. If one spore germinates into unfavorable conditions and fails, others remain dormant, preserving the genetic lineage. It ensures the fungus doesn't put all its eggs in one basket.
The Germination Paradox: Does Clustering Help or Hinder?
This is the central question. The answer is nuanced: clustering delays but optimizes germination.
- The Delay: The physical barrier of the crust and the need to re-hydrate a larger, denser structure mean that clustered spores often germinate more slowly than solitary spores. They require a stronger, more persistent stimulus (like continuous root exudates) to initiate the process.
- The Ultimate Advantage: This delay is strategic. It acts as a dormancy filter, ensuring germination only occurs when conditions are truly favourable and a host root is reliably nearby. When they do germinate, they benefit from the "synergistic signals" and pre-built hyphal connections, potentially leading to more robust and successful colonization. The cluster ensures quality over speed.
A Call to Action: Why Specialists MUST Reconsider Spore Counting
This brings us to a critical methodological point. For mycorrhizal ecologists, agronomists, and inoculum producers, treating a spore cluster as "one" is a significant error. Here’s why:
- Inaccurate Propagule Potential: A cluster of 10 spores has 10x the genetic potential and nutrient reserves of a single spore. Counting it as one propagule drastically underestimates the true infectious potential of the soil or inoculum.
- Skewed Biodiversity & Abundance Data: In ecological surveys, failing to dis-aggregate clusters lead to severe underestimation of spore density and can distort comparisons between species (some cluster more than others) or between management practices.
- Misjudging Inoculum Quality: The proportion of clustered vs. solitary spores in a commercial inoculum could be a key quality indicator. A high cluster ratio suggests a product designed for resilience and long-term survival, while a high solitary spore count might indicate faster, but less persistent, colonization.
Best Practice: The specialist's approach should involve gentle crushing of samples in a weak surfactant solution to break up clusters before counting, providing a true measure of total spore numbers. Documenting the degree of clustering separately adds a valuable layer of ecological insight.
“Taken together, the physical structure, biochemical signals, and germination patterns indicate that the spore cluster functions as a unified survival module.".
Clustering represent a shift in how we view these fungi—not as collections of individual spores, but as complex, cooperative societies engineered for persistence.
Their sticky glue is the mortar of a survival fortress, their delayed germination a mark of wisdom, and their continued underestimation in counts a gap in our understanding.
By acknowledging and studying these clustered formations in their full complexity, we unlock a deeper appreciation of mycorrhizal ecology and move closer to harnessing their full power for sustainable agriculture and ecosystem restoration.
“Look closer. Count smarter. The future of soil health depends on such microscopic details.”
Golden Is Good… But So Are White and Pale Yellow Spores—Here’s the Science
A Story from the Farm That Opened Everyone’s Eyes
A farmer once visited our field demo carrying two samples of mycorrhiza spores.
One had golden-yellow spores—bright, shiny, and visually impressive.
The other had white and pale-yellow spores—simple, dull, easy to underestimate under microscopy.
He asked:
“Saheb, aa banema thi kayu sachu kaam karse? (Which one will work better?)”
Almost everyone pointed to the golden spores.
But the soil told a different story!
When we tested both samples:
- Some golden spores did not stain in the MTT assay → meaning they were dormant or old
- The white and pale-yellow spores turned deep blue hue→ showing strong viability and active metabolism
- Pale spores colonized roots more quickly
- Field plots with pale spores showed better early growth and stronger root networks
The farmer was surprised.
But this is exactly what the science says: Golden spores are good—but white and pale-yellow spores are equally good, often even more active.
Why Colour Alone Misleads Us
A mycorrhiza spore’s colour—whether white, pale yellow, cream, or golden—naturally varies due to its genetics, the host plant it grew with, the soil and environment it developed in, its age, and the carotenoid pigments in its outer wall. Because these factors influence only the outer appearance, colour alone cannot indicate a spore’s internal health or viability. Just like wheat grains that differ slightly in shade but grow equally well, mycorrhiza spores also show harmless colour variation. The real indicator of quality is not colour, but whether the spore is alive and capable of colonizing roots.
White and Pale-Yellow Spores: Naturally Efficient Contributors to Soil Health
White and pale-yellow AMF spores are not weak or immature—multiple authoritative sources, including INVAM (International Culture Collection of Arbuscular Mycorrhizal Fungi) and GINCO-BEL (Belgian Glomeromycota Collection), document that many well-known, highly functional AMF species naturally produce spores in lighter shades. These pale-coloured spores belong to species that play a strong role in nutrient mobilization, hyphal spread, soil aggregation, and root colonization, often performing as effectively as darker or golden spores. AMF reference databases also show clear evidence that pale-coloured spores are common in Glomeraceae and other agriculturally important families, where colour variation is considered a normal taxonomic trait, not a sign of poor quality.
Because of this natural diversity (Polymorphism), pale and white spores consistently contribute to soil ecology by improving phosphorus and micronutrient uptake, enhancing water-use efficiency, increasing soil microbial activity, and supporting carbon storage through extensive hyphal networks.
Thus, the presence of white or pale spores in a product is fully aligned with natural AMF biology and reflects the diversity found in verified, curated AMF collections worldwide.
Myth vs. Reality: Colour Does Not Indicate Viability
It is a common belief that yellow or golden spores are always viable and white or pale spores are non-viable, but MTT assays and global AMF references show this is not true. Golden spores often fail to stain in MTT because they may be senescent, dormant, or pigmented in a way that limits dye penetration, meaning their colour alone cannot confirm activity or viability. In contrast, white and pale-yellow spores frequently stain deep blue, indicating active metabolism, healthy cytoplasm, and readiness to germinate.
So the reality is clear:
Yellow spores are not automatically viable, and white/pale spores are not automatically non-viable. Viability depends on the internal physiology of the spore, not its colour.
What Actually Determines Spore Quality?
Real quality depends on:
- Germination ability
- Cytoplasmic health
- MTT viability
- Hyphal growth
- Root colonization %
- Infection points
- Field performance
Not on colour.
Colour only tells appearance—not power.
Appearance Isn’t Quality: Rethinking AMF Procurement Standards
For procurement teams, judging spore quality by colour alone may seem simple, but it causes significant hidden losses. When white or pale-yellow spores are rejected purely because they don’t “look” ideal, procurement unknowingly discards many viable, high-performing spores that global AMF collections such as INVAM and GINCO-BEL consider completely natural. This leads to avoidable batch rejections, reduced usable output, higher production costs, and inconsistent final product quality.
Colour-based filtering also increases the risk of supplying farmers with visually appealing—but biologically weaker—products. By removing pale spores that often show strong metabolic activity in tests like MTT, procurement unintentionally reduces the overall viability of the formulation. This undermines field performance and long-term trust in the brand.
Moreover, rejecting pale spores deprives the soil of beneficial fungi that contribute to nutrient uptake, root development, and soil structure. These losses highlight why global AMF experts do not use colour as a quality parameter. Procurement accuracy improves dramatically when decisions are based on functional viability, germination, and colonization ability, rather than superficial appearance.
What Should Be Practical Action Check List?
1. Update Procurement SOPs
Adopt the natural colour range of AMF spores — white, cream, pale yellow, and light golden — as acceptable and normal variations.
2. Train QC & Field Teams
Clarify that lighter spores are not weak; they are natural and often show higher metabolic activity in viability tests.
3. Educate Farmers in Simple Terms
Use farmer-friendly explanations such as:
“Spore nu rang quality nathi batavtu; MTT ma neelo rang jivant spore batave chhe.”
4. Follow Science-Based Quality Checks
Rely on MTT viability, germination, and colonization efficiency, rather than colour-based selection.
5. Promote Soil-Health–First Communication
Show how pale spores improve nutrient uptake, root strength, and long-term soil fertility year after year.
Spore Colour Is Just Nature; Viability Is the Truth
Golden spores are good.
White spores are good.
Pale-yellow spores are good.
Every colour is simply a part of nature’s diversity—and all can support strong roots, healthy soil, and better crops. What truly matters is viability, not appearance.
It’s time to move beyond colour myths and build a future driven by science, soil health, and honest quality standards that deliver real value to farmers.
Because ultimately…
Crops don’t grow from the colour of the spore—they grow from the life and activity within it.

Small but Mature! The Difference Between Natural Size and Immaturity in AMF Spores
Why spore size alone can’t tell you the whole story?
Have you ever bitten into a fruit too early? A mango that’s still hard and sour, or a banana that’s green and starchy? You know instantly—it wasn’t ready. But what about cherries or berries? They’re small, yet perfectly sweet when ripe.
AMF (Arbuscular Mycorrhizal Fungi) spores work a lot like fruits. Some are naturally small but ready for action, while others are small because they’re still growing. The real trick is knowing the difference.
Small by Nature vs. Small by Immaturity
Small by Nature
Just like cherries are small but ripe, some AMF species (Glomus clarum, for example) always make small spores—60–100 µm in size. They may be tiny, but they’re complete, mature, and capable of doing their job.
Small by Immaturity
Other species (Gigaspora gigantea) are designed to grow big—up to 400 µm. If you find one of its spores at only 100 µm, it’s like biting into a half-grown mango. It hasn’t yet packed in the nutrients, energy reserves, or protective walls it needs. In short: it’s unfinished.
The Spore’s “Coming of Age” Story
Every spore goes on a journey, almost like a child growing into an adult:
- The Baby Stage – A little swelling appears at the fungal tip.
- The Teenager – It grows rapidly, storing lipids and sugars, bulking up, but still fragile.
- The Young Adult – Its wall hardens into multiple protective layers.
- The Independent Adult – It cuts ties from its fungal parent and becomes its own strong, viable unit.
If you find it in the teenage stage—it looks smaller, softer, and isn’t ready to take on the world.
Signs of an Immature Spore
Under a microscope, immature spores reveal their secrets:
- Smaller than the normal size of its species.
- Thin or translucent wall.
- Watery cytoplasm with few oil droplets.
- Still attached to the fungal parent.
- Cannot germinate properly.
Why Does This Matter?
- For Science: Avoid inflated spore counts.
- For Farmers: Only mature spores actually help crops.
- For Ecology: A wave of immature spores often means fungi are actively reproducing, usually after rain or fresh carbon from roots.
The Big Picture
Not every small thing is incomplete. Some spores are born small and strong. Others are only temporarily small because they’re still developing. Just like fruit, the difference between “ripe” and “unripe” changes everything.
- For researchers, this means accurate counts.
- For producers, it means honest quality control.
- For farmers, it means inoculum you can truly trust.
In the end: it’s not the size of the spore that matters—it’s whether it’s ready for the job.
Farmer’s Analogy Box: Spores = Fruits
- Cherries (Small but Ripe) → Naturally small spores (Glomus clarum) → Mature and ready.
- Mangoes (Big, Unripe when Small) → Immature spores (Gigaspora gigantea) → Still developing.